Description
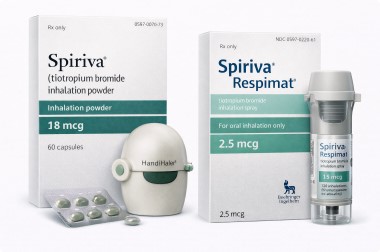
Spiriva is a prescription maintenance treatment containing tiotropium and is available in two professional formats: Spiriva 18 MCG and Respimat 2.5 MCG. Both options are designed for once-daily maintenance use and are recognized treatment choices in long-term respiratory care.
Spiriva 18 MCG is supplied as capsules for use with the HandiHaler device, and the current FDA label states it is indicated for the long-term, once-daily maintenance treatment of bronchospasm associated with COPD, including chronic bronchitis and emphysema, and to reduce exacerbations in COPD patients.
Respimat 2.5 MCG is supplied as an inhalation spray. FDA labeling states that each actuation delivers 2.5 mcg of tiotropium, with two inhalations equal one dose, and the recommended COPD dose is two inhalations once daily for a total daily dose of 5 mcg.
Why Choose Spiriva?
Spiriva offers two convenient maintenance options to match different patient preferences and inhalation formats.18 MCG provides a capsule-based HandiHaler option, while Respimat 2.5 MCG offers a soft mist inhaler format for once-daily use. Both are established tiotropium maintenance therapies used in professional respiratory care.
Common Usage & Available Strengths
Spiriva is available as 18 MCG and Respimat 2.5 MCG.18 MCG is used with the HandiHaler device for once-daily maintenance treatment in COPD.Respimat 2.5 MCG is an inhalation spray where two inhalations equal one dose, for a total daily dose of 5 mcg in COPD. The appropriate product type and dosing schedule should always follow the recommendation of a licensed healthcare provider.










Reviews
There are no reviews yet.